Sustanon 250: Uses, Legal Status & Availability in Canada, Sweden, France & Germany
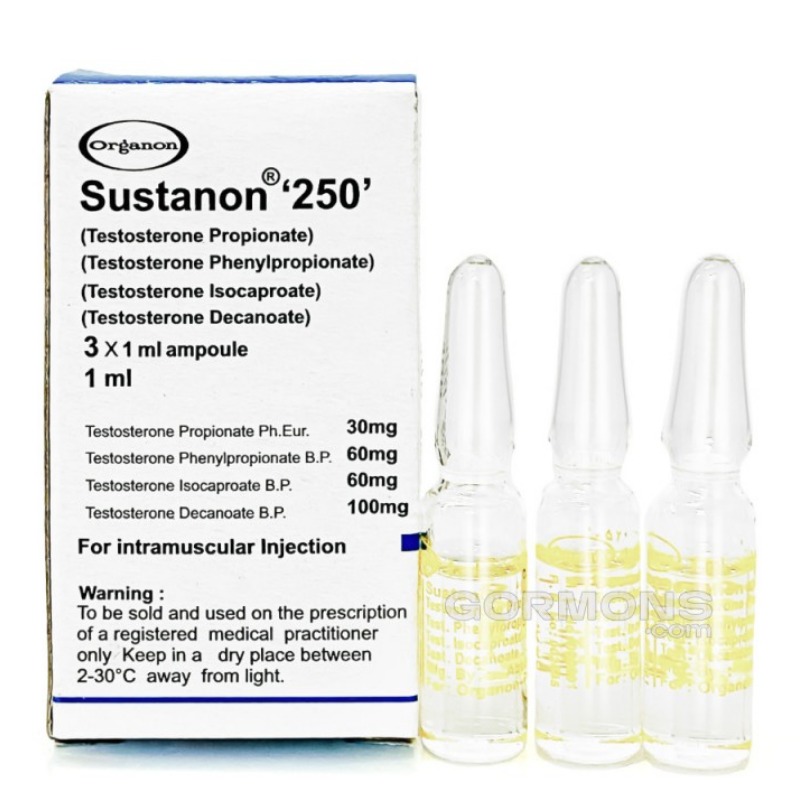
Sustanon 250 is a widely prescribed long-acting injectable testosterone blend used in testosterone replacement therapy (TRT). It contains four different testosterone esters designed to provide both rapid onset and sustained hormone release, making it popular among clinicians for managing male hypogonadism.
This guide explains the availability, regulation, and medical use of Sustanon in Canada, Sweden, France, and Germany.
What is Sustanon 250?
Sustanon is a combination of four testosterone esters:
Testosterone Propionate
Testosterone Phenylpropionate
Testosterone Isocaproate
Testosterone Decanoate
This ester blend allows:
Faster initial testosterone rise
Prolonged hormonal stability
Reduced injection frequency (every 2–4 weeks in clinical use)
Medical Uses of Sustanon
Sustanon is prescribed for:
Male hypogonadism
Testosterone deficiency due to testicular or pituitary disorders
Hormone replacement therapy under specialist care
It is intended strictly for prescription-based medical use.
Availability in Canada
In Canada, Sustanon is regulated by Health Canada and is prescription-only.
Availability:
Endocrinologists and urologists
Hospital and retail pharmacies
Licensed hormone therapy clinics
Testosterone products are classified as controlled substances and require strict compliance.
Availability in Sweden
Sweden regulates Sustanon through the Swedish Medical Products Agency (Läkemedelsverket).
Key points:
Prescription-only medicine
Commonly prescribed in public healthcare
Dispensed mainly via hospital or specialist clinics
Sweden has strict hormone monitoring protocols.
Availability in France
In France, Sustanon is regulated by ANSM (Agence nationale de sécurité du médicament).
Availability includes:
Specialist endocrinology prescriptions
Hospital pharmacies
Approved private prescriptions
France enforces strong pharmacovigilance for androgen therapies.
Availability in Germany
Germany regulates Sustanon under BfArM (Federal Institute for Drugs and Medical Devices).
It is:
Prescription-only
Available through licensed pharmacies
Commonly used in TRT compared to single-ester testosterones
German guidelines emphasize long-term monitoring and dosage control.
Storage & Handling
Store at 20°C–25°C (68°F–77°F)
Do not refrigerate unless specified
Protect from light
Administer via intramuscular injection only
Safety & Compliance Notice
Sustanon should only be used under medical supervision. Regular blood tests are required to monitor testosterone levels, hematocrit, and overall hormonal balance.
📞 Contact Global Meds Export
Global Meds Export – Delivering Hormone Therapies with Compliance & Care
📧 Email: ritecareind@gmail.com
📲 WhatsApp: +91 91685 08880
🌐 Website: www.globalmedsexport.com